TNF Inhibitor Risk & Screening Guide
Select your prescribed medication and current screening status to understand your risk profile and the recommended medical next steps.
Clinical Analysis
The risk isn't the same for every drug in this class. Depending on which medication you're prescribed, your risk levels can shift dramatically. Most of these cases happen within the first three to six months of starting therapy, making the initial screening window the most critical part of your care plan. If you're about to start these meds, or you're already on them, understanding how to spot the warning signs and why the screening process is so rigorous can literally save your life.
Why some TNF inhibitors are riskier than others
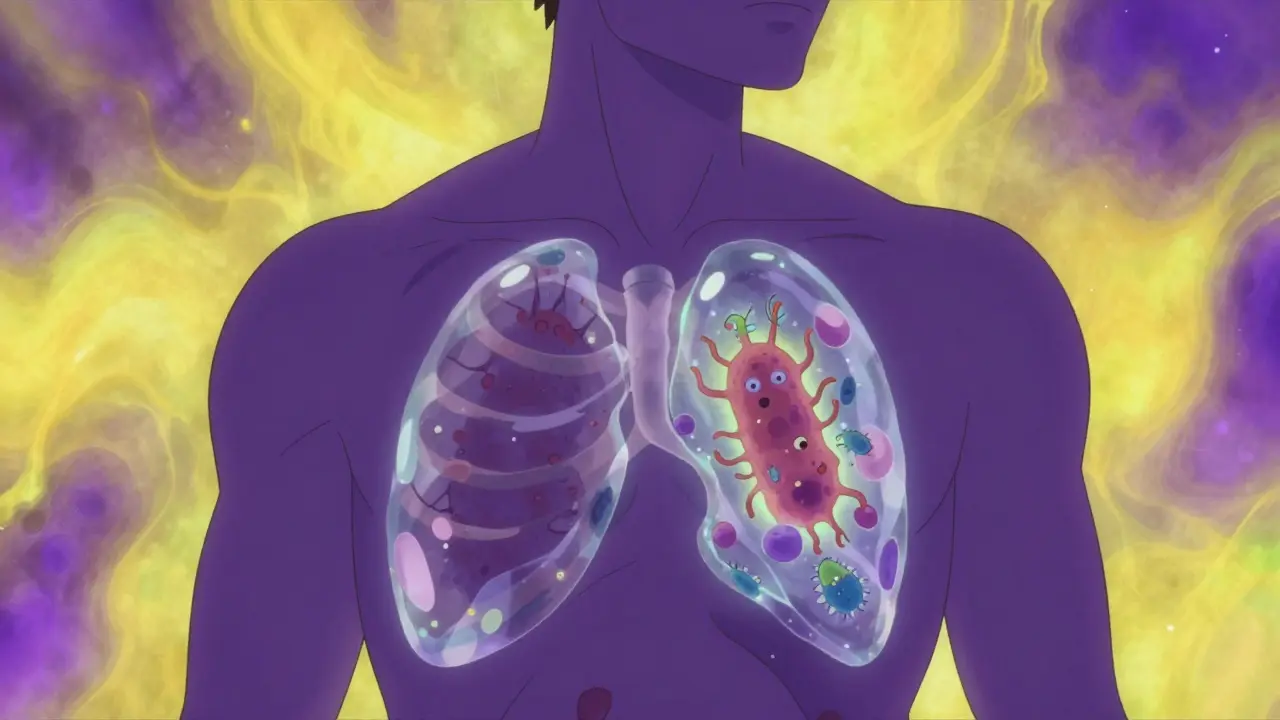
Not all TNF blockers work the same way. To understand why some cause more TB cases than others, you have to look at how they interact with the cells in your body. Your immune system traps TB bacteria inside tiny, wall-like structures called granulomas. Tumor necrosis factor-alpha (TNF-α) is the "glue" that keeps these walls intact. If the glue fails, the bacteria escape and the infection spreads.
There are two main types of inhibitors here. First, you have soluble receptor decoys like etanercept. These mostly soak up the floating TNF-α in your bloodstream. Because they leave the membrane-bound TNF (the glue on the cell walls) mostly alone, the risk of TB reactivation is significantly lower. In some studies, the risk with etanercept was less than a quarter of what was seen with other blockers.
Then you have the monoclonal antibodies, such as adalimumab and infliximab. These are more aggressive; they bind to both the floating and the membrane-bound TNF. By stripping the glue directly off the cell walls, they effectively dismantle the granulomas, giving the TB bacteria a free pass to enter the bloodstream. This is why patients on these specific drugs often see a three-fold increase in TB incidence compared to those on etanercept.
| Drug Name | Mechanism | TB Risk Level | Impact on Granulomas |
|---|---|---|---|
| Etanercept | Soluble Receptor | Lower | Preserves membrane-bound TNF |
| Adalimumab | Monoclonal Antibody | Higher | Disrupts membrane-bound TNF |
| Infliximab | Monoclonal Antibody | Higher | Disrupts membrane-bound TNF |
The screening process: How we find the hidden threat
Since you can't "feel" a latent infection, doctors have to hunt for it before the first dose of biologic therapy hits your system. There are two main tools for this: the tuberculin skin test (TST) and the interferon-gamma release assay (IGRA). The TST is the old-school method involving a small injection under the skin, while the IGRA is a modern blood test.
Here is the typical workflow for a patient starting TNF inhibitors:
- Initial Screen: A blood test (IGRA) or skin test (TST) is performed to see if the body recognizes TB proteins.
- Risk Assessment: The doctor looks at where you've lived. If you're from a high-burden country (more than 40 cases per 100k people), the rules change; you might get treated even with a negative test.
- Preventative Treatment: If you test positive for LTBI, you start a course of antibiotics-usually isoniazid-for several months. You typically need at least one month of this treatment before the TNF inhibitor is introduced.
- Verification: Ensuring the preventative meds aren't causing liver issues (hepatotoxicity) before proceeding.
It's worth noting that screening isn't a perfect shield. Real-world data shows that about 18% of people who actually develop TB while on these drugs had negative tests at the start. This happens because the test might miss a very small amount of bacteria, or the patient might have been infected just before starting therapy.

Monitoring during therapy: What to watch for
Screening is just the start. Once you're on a biologic, you need a long-term monitoring strategy. Most guidelines suggest a quarterly check-in for the first year, and then an annual review after that. But you shouldn't wait for your appointment if you notice specific red flags.
Unlike a standard chest cold, TB reactivation under immunosuppressants can look "weird." In the general population, TB usually hits the lungs. However, in people on TNF blockers, about 78% of cases show extrapulmonary involvement. This means the infection might show up in your lymph nodes, liver, or even your brain, rather than just causing a cough.
Keep an eye out for these systemic symptoms:
- Unexplained fever or persistent low-grade chills.
- Drenching night sweats that require you to change your sheets.
- Unintentional weight loss, even if your appetite is normal.
- A chronic cough that doesn't respond to standard cold medicine.
If you're treated for TB while on these meds, be aware of something called TB-IRIS (Immune Reconstitution Inflammatory Syndrome). This happens when your immune system suddenly "wakes up" and attacks the TB bacteria with such intensity that it causes massive inflammation. It often happens within 45 days of starting TB treatment and can require high doses of steroids to keep the inflammation under control.

Practical challenges and modern solutions
The biggest hurdle in the clinic isn't the science-it's the adherence. The traditional 9-month isoniazid regimen is grueling. Many patients quit halfway through because of nausea or concerns about liver damage. To fix this, newer guidelines have introduced a 4-month rifampin/isoniazid combination. Clinical trials show this shorter window boosts adherence from 68% to 89%, making it much more likely that patients actually finish their preventative therapy.
Another challenge is the "false negative" anxiety. In high-risk populations, clinicians are increasingly moving toward a two-step screening process: starting with an IGRA and following up with a TST if the first is negative. This layering increases the chance of catching a latent infection that a single test might have missed.
Looking ahead, the medical community is working on "selective" TNF inhibitors. These are designed to target only the floating TNF-α and ignore the membrane-bound version. Early animal models suggest these could reduce the risk of TB reactivation by up to 80%, potentially removing the need for some of the more aggressive screening protocols we use today.
Can I start a TNF inhibitor if my TB test is positive?
Yes, but not immediately. If you test positive for latent TB, you must start preventative antibiotic treatment first. Most doctors require at least one month of this treatment before the first biologic dose is administered to ensure the bacteria are suppressed.
Why is the IGRA blood test preferred over the skin test?
The IGRA test is generally more specific. The skin test (TST) can sometimes give a "false positive" if you've had the BCG vaccine for TB, whereas the IGRA blood test can distinguish between the vaccine and an actual latent infection.
What should I do if I develop a cough while on Adalimumab?
Contact your rheumatologist or primary care doctor immediately. Because TNF inhibitors mask some immune responses, a simple cough could be the first sign of reactivation. Mention specifically that you are on a TNF blocker so they can prioritize a chest X-ray or sputum test.
Is etanercept truly safer regarding TB?
Statistically, yes. Because etanercept doesn't bind as strongly to the membrane-bound TNF that maintains the granuloma "wall," the risk of the bacteria escaping is lower compared to monoclonal antibodies like infliximab.
How long does the risk of reactivation last?
While the risk is highest in the first 3-6 months, you are never completely "clear" as long as you are on the medication. Regular annual screening and symptom monitoring are recommended for the entire duration of your therapy.
Next steps for patients and caregivers
If you are starting a new biologic, ask your doctor for a written copy of your screening results. Knowing whether you had a positive or negative IGRA/TST will be helpful if you ever change providers or need emergency care. If you are from a region with high TB prevalence, don't be afraid to ask for the two-step screening process to be sure.
For those already on therapy, set a calendar reminder for your annual check-up. If you notice a pattern of night sweats or unexplained weight loss, don't dismiss it as "just getting older" or "stress." In the context of TNF inhibitors, these are clinical markers that require immediate medical investigation.

dwight koyner
April 8, 2026 AT 07:01The distinction between soluble receptors and monoclonal antibodies is a crucial point for patient education. Many individuals are unaware that the mechanism of action directly influences the risk of granuloma disruption, which is the primary driver of TB reactivation in these cases.
Christopher Cooper
April 9, 2026 AT 22:13It's fascinating how specific the targeting has to be to avoid these side effects. I wonder if these newer selective inhibitors will eventually become the gold standard for everyone starting biologic therapy to minimize the anxiety around screening.
Daniel Trezub
April 10, 2026 AT 08:54Pretty basic stuff here. Everyone knows the TST is outdated, but the post acts like it's some huge revelation. Also, calling etanercept "safer" is a bit of a stretch when you consider the overall efficacy for certain conditions compared to adalimumab. Just my two cents!
Benjamin cusden
April 11, 2026 AT 03:17The analysis is elementary. One should actually understand the molecular biology of cytokine signaling before attempting to categorize these risks. The nuance regarding membrane-bound TNF is well-documented in academic literature, though I suppose for a general audience, this simplified table suffices.
Ethan Davis
April 13, 2026 AT 00:48Sure, they tell you the risk is "low" with some drugs, but why are they really pushing these biologics? It feels like a way to get people on expensive meds while ignoring the long-term damage to the immune system. Who knows what else they're waking up inside us!
Michael Flückiger
April 14, 2026 AT 04:59Just stay positive!!! If you're going through the screening, just keep pushing through it!!! It's all for your own good in the end!!!
jack hunter
April 14, 2026 AT 05:20whatever... health is just a slow decay anyway. why screen for one thing when the whole system is just failng? its all just a cycle of meds to fix medes. totaly pointless if u think about the big picture of existance.
Sarabjeet Singh
April 16, 2026 AT 04:48Good to know.
Jamar Taylor
April 17, 2026 AT 16:17For anyone feeling overwhelmed by the 9-month antibiotic grind, remember that the shorter 4-month rifampin option is out there now. Just keep talking to your doctor and stay on track with your treatment. You've got this, and the effort is totally worth the peace of mind and the relief from your joint pain. It's a marathon, not a sprint, but the finish line is a healthier you. Just keep focusing on the small wins every day. Each dose of preventative medicine is a step toward safety. Don't let the nausea get you down; the reward is a life without those painful flares. Stay strong and keep your eyes on the goal. You are doing the right thing for your body. Just trust the process and keep moving forward. One day at a time, and you'll get through the screening and into the treatment that works for you. Keep that energy up!